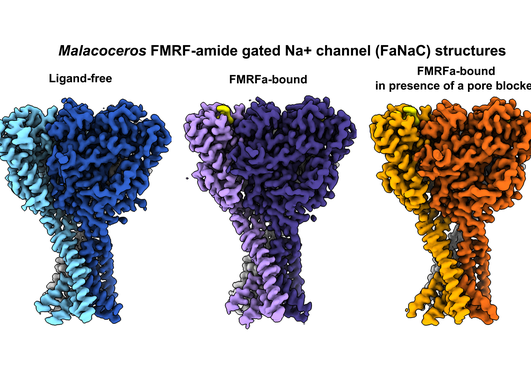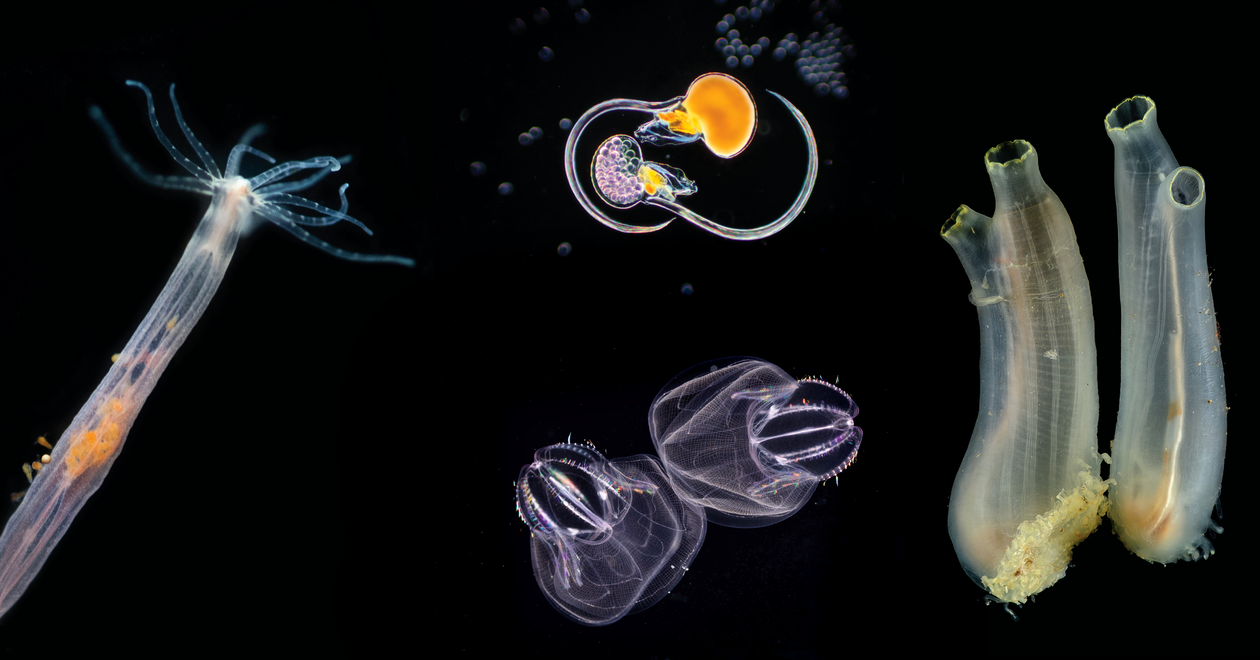
The Michael Sars Centre at the University of Bergen, is an international community of scientists using advanced technologies to study the unique molecular and cellular biology of marine organisms in a changing environment for broad societal impact.
As one of the first EMBL partners, the Michael Sars Centre is rooted in the Bergen academic community and serves as a national strategical asset for Norwegian marine life sciences. We aim to establish, strengthen, and leverage local, national, and international networks through specific activities, including collaborative research, joint training, and scientific exchange.
Calendar
Dr. Nina Cabezas-Wallscheid, Max Planck Institute of Immunobiology and Epigenetics, Germany
Michael Sars Seminar Room. Thormøhlensgt. 55, 5006 Bergen
26.04.2024
10:00–11:00
Sars Seminar Room. Thormøhlensgt. 55, 5006 Bergen
Trial Lecture: Yuhong Wang (Trial lecture)
13:15–14:15
Sars Seminar Room. Thormøhlensgt. 55, 5006 Bergen
Dr. Kristina Noreikiene, Estonian University of Life Sciences (Seminar)
10:00–11:00
Michael Sars Seminar Room. Thormøhlensgt. 55, 5006 Bergen
Professor Frank Conlon, University of North Carolina, USA. (Seminar)
11:00–12:00
Doctoral Dissertation Defense: Yuhong Wang (Disputation)
10:00–13:00